What is a Concentration Gradient in Biology?
Are you having difficulty understanding the concept of concentration gradient in IGCSE/O Level Biology. If you find yourself in the same boat, worry not! We have prepared this quick O Level/IGCSE concept guide just for you!
Definition of Concentration Gradient
As the name suggests, concentration gradient in Biology refers to the gradual change in concentration of particles.
Example of Concentration Gradient in Biology
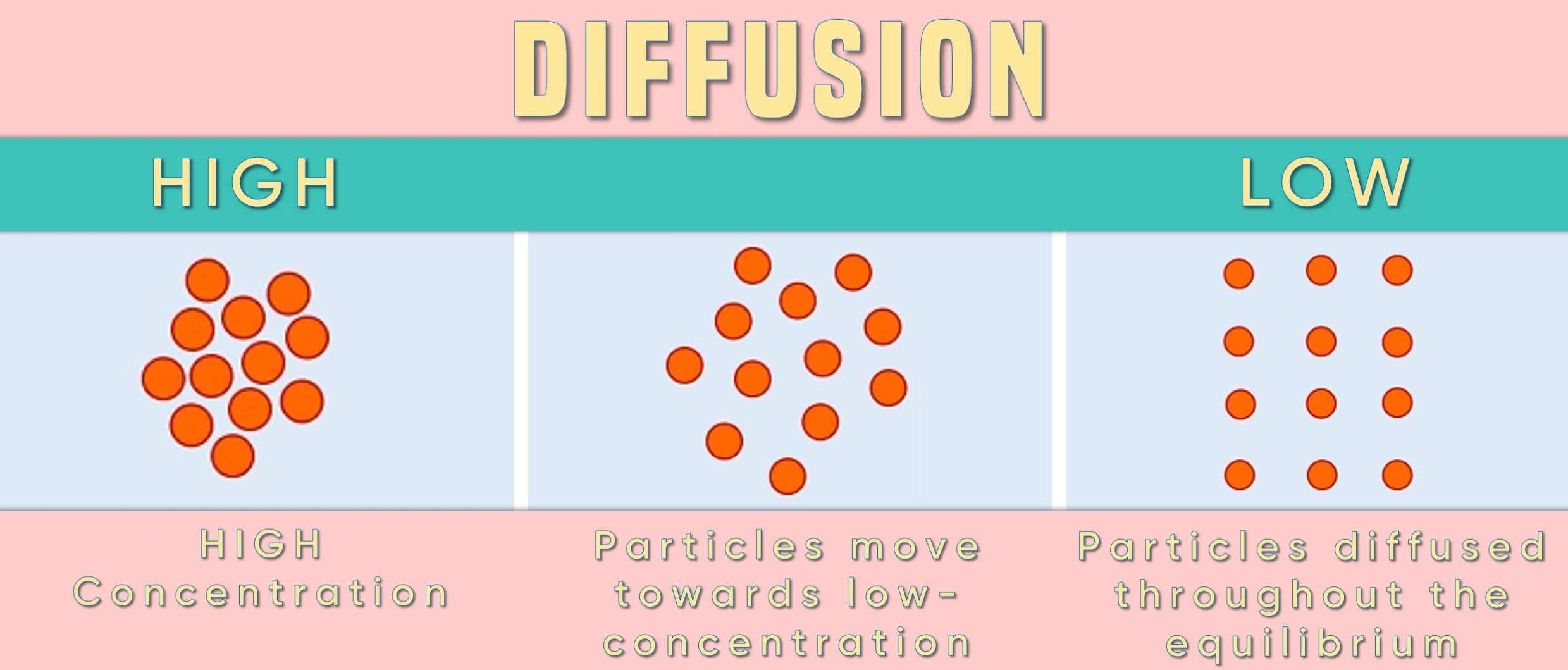
If you place a drop of food colouring in water, a concentration gradient will be set up.
At the start:
-
The food colouring particles will be high in concentration where the initial drop is placed
-
Food colouring particles will be low in concentration and far away from the drop
-
Observe that there will be a decrease in the concentration of food colouring particles as you go away from the drop.
This difference in the concentration of particles at the various points creates the concentration “gradient”.
Why is Concentration Gradient Important?
Concentration gradients enable diffusion in biological systems. For example, oxygen diffuses into cells from the bloodstream and the concentration gradient across the cell membrane enables this.
Does Diffusion Require a Concentration Gradient?
Yes, certainly! Diffusion is the flow of particles from a region of high concentration to a region of lower concentration. The concentration gradient provides this “slope/gradient” of concentration. Hence, particles flow “down” the concentration gradient.
How does the concentration gradient affect diffusion?
The greater the concentration gradient the faster the diffusion rate.
How is a Concentration Gradient Maintained?
The concentration gradient is maintained through:
-
Active transport: involves pumping molecules against the flow to maintain a concentration gradient in one direction.
-
Metabolic processes: In respiration, oxygen is consumed within the cell. This ensures a lower oxygen concentration inside the cell compared to the bloodstream.
Summary
In conclusion, understanding “what is a concentration gradient” is fundamental in biology.
-
Concentration gradient is vital for biological diffusion
-
Differences in particle concentration establish it
-
It allows substances like oxygen to move across cells
If you found this guide helpful, you can stay tuned for more articles on O Level/IGCSE Biology from Out-Class.
Most Common Repeated Questions:
Unlock the secrets to acing your CAIE IGCSE & O Level exams with a sneak peek into the most frequently asked questions that have graced the past papers!
- The roots of the plants are provided with a nutrient solution. The nutrient solution contains magnesium ions. Air is pumped through the nutrient solution using an air stone that contains many very small holes. State the process that transports magnesium ions against a concentration gradient into this type of cell. (1) [Oct/Nov 2020]
FAQs:
Q. What is a concentration gradient in Biology?
Concentration gradient refers to the gradual change in the concentration of particles. It represents the difference in particle concentration between two points.
Q. Can you provide an example of a concentration gradient?
Yes, if you place a drop of food coloring in water, a concentration gradient will be set up. Initially, the food coloring particles are highly concentrated near the drop, and the concentration decreases as you move away.
Q. Does diffusion require a concentration gradient?
Yes, diffusion involves the flow of particles from a region of high concentration to a region of lower concentration. The concentration gradient provides the slope for this flow.
Q. How does the concentration gradient affect the diffusion rate?
The greater the concentration gradient, the faster the diffusion rate. A steeper concentration gradient results in a more rapid movement of particles.
Q. What are some practical examples of concentration gradients in biological processes?
Practical examples include the diffusion of oxygen from the bloodstream into cells, the movement of ions across cell membranes, and the distribution of molecules within cellular structures.